Left-click: rotate, Mouse-wheel/middle-click: zoom, Right-click: pan, Escape: close
Fluorescence
- Conversion of UV to visible light.
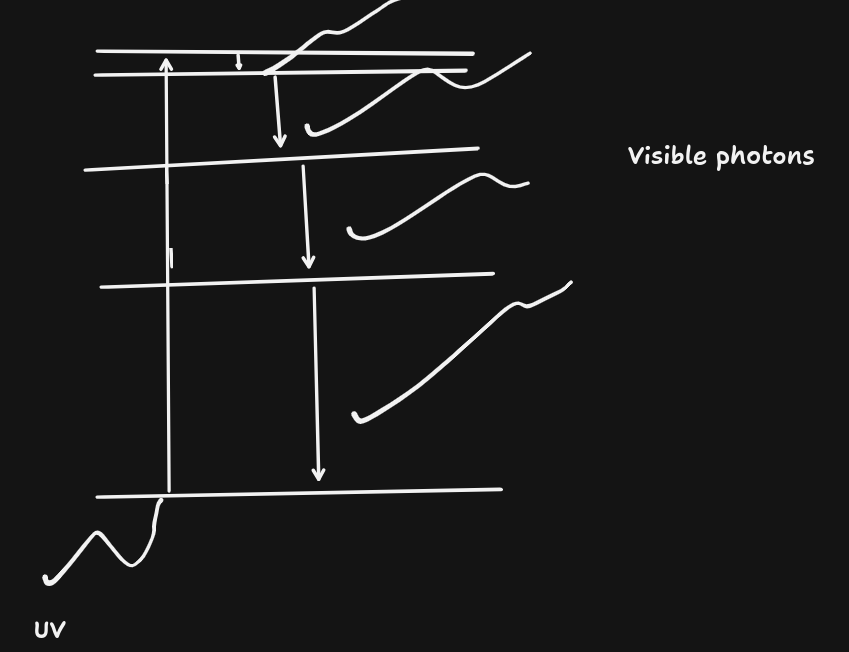
- e.g. Whitening agents in laundry powder, teeth whitening!
"Seems like light is coming out from where there was no light" (Because UV isn't visible)
- Usually white light is produced.
Gas discharge lights:
- Mercury vapour and high voltage produces high-energy photons (UV)
- UV hits fluorescent paint on the surface, producing white visible light.
Phosphorescence
Each exited electron is metastable. This means they release photons later on.
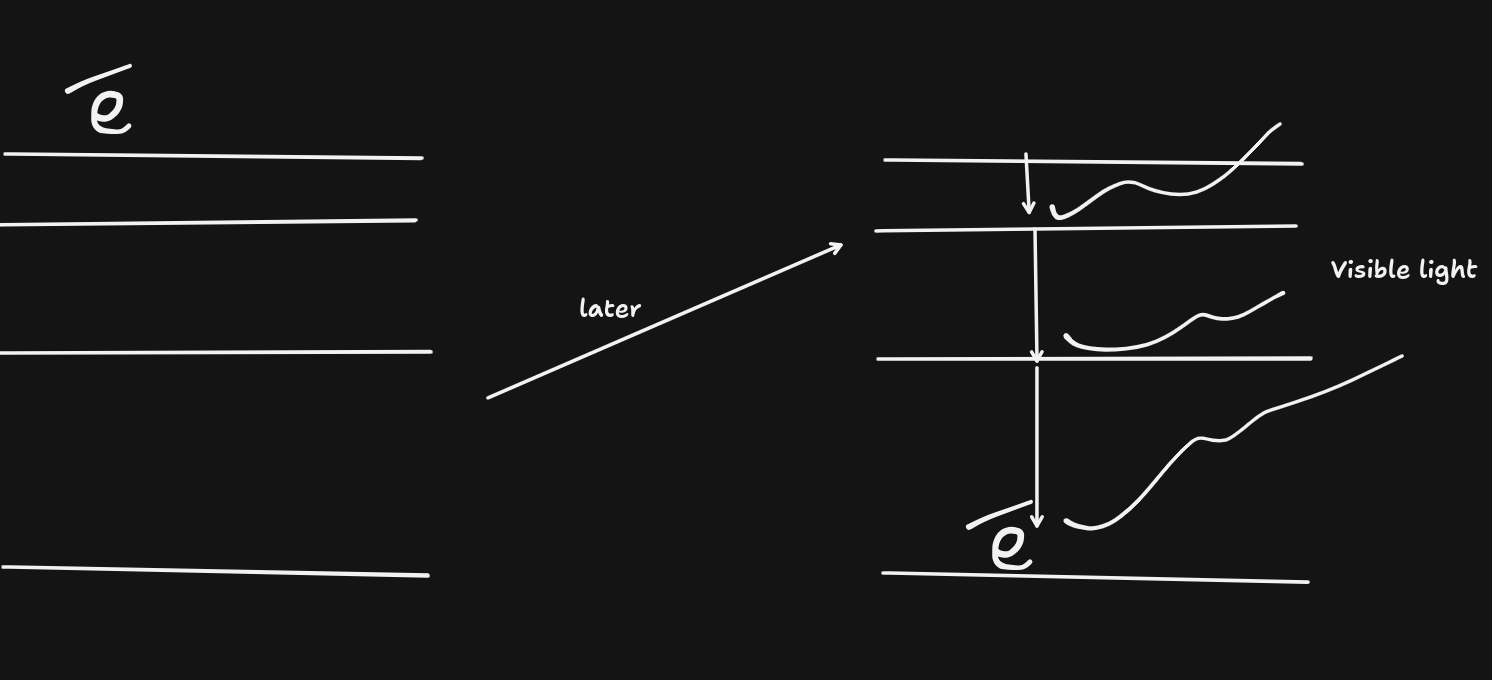 (Glow in the dark)(luminous)
(Glow in the dark)(luminous)
LASER
Energy drops produce photons all at the same frequency, resulting in the 'laser'.

Exciting electrons
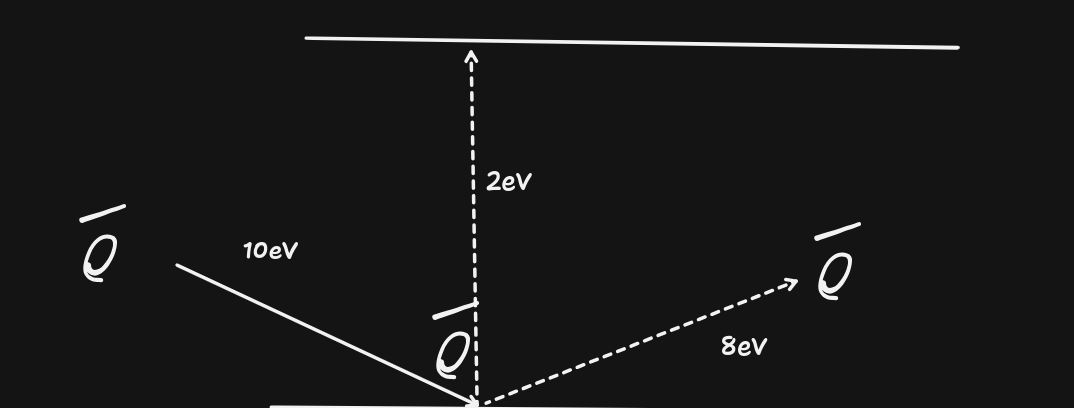
- Absorption of photon - must be absorbed fully, i.e. all the energy given to electron. Must also have the exact energy equal to discrete differences in energy levels(unless ionising, remaining energy is kinetic of electron).
- Electron bombardment (scattering) - electron can deliver some of its energy another electron, and move off with less $E_{k}$.
LED - Light emitting diodes
- Similar to laser. A certain energy drop for excited electrons corresponds to the frequency of light which is emitted.

 (Glow in the dark)(luminous)
(Glow in the dark)(luminous)
