Left-click: rotate, Mouse-wheel/middle-click: zoom, Right-click: pan, Escape: close
Young's double slit experiment
-
If light acted as a particle then it would produce 2 slits
-
But if light acts as a wave then it produces many bands.
-
$c=3\times 10^8ms^-1$ - speed of light
-
$c=f\lambda$
Photoelectric effect - uh oh waves
- Thought about by Einstein and Planck (Einstein got his Nobel prize for this!)
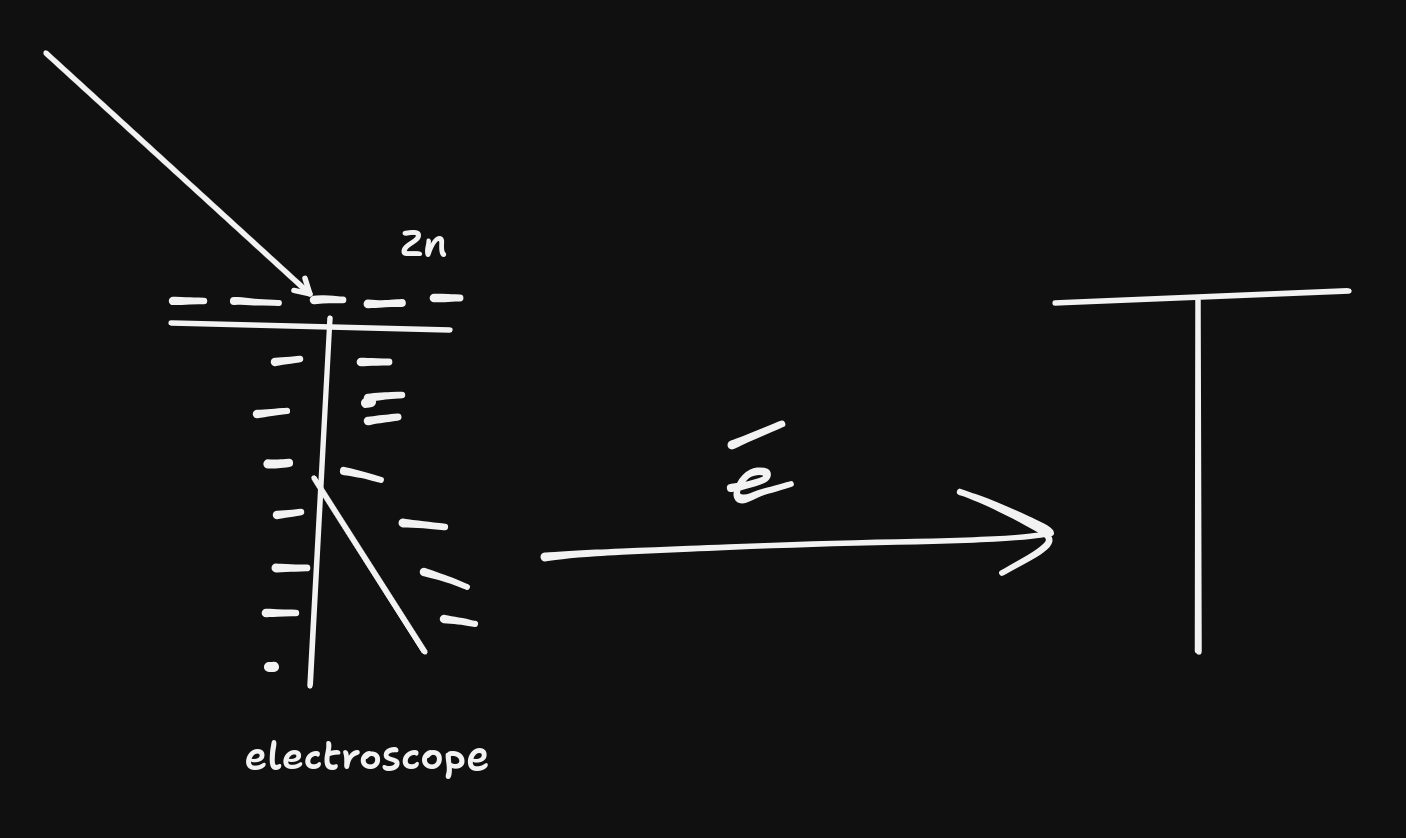
- The inference made is that the UV light was kicking electrons off the $Zn$ plate.
- The top left arrow is UV light btw
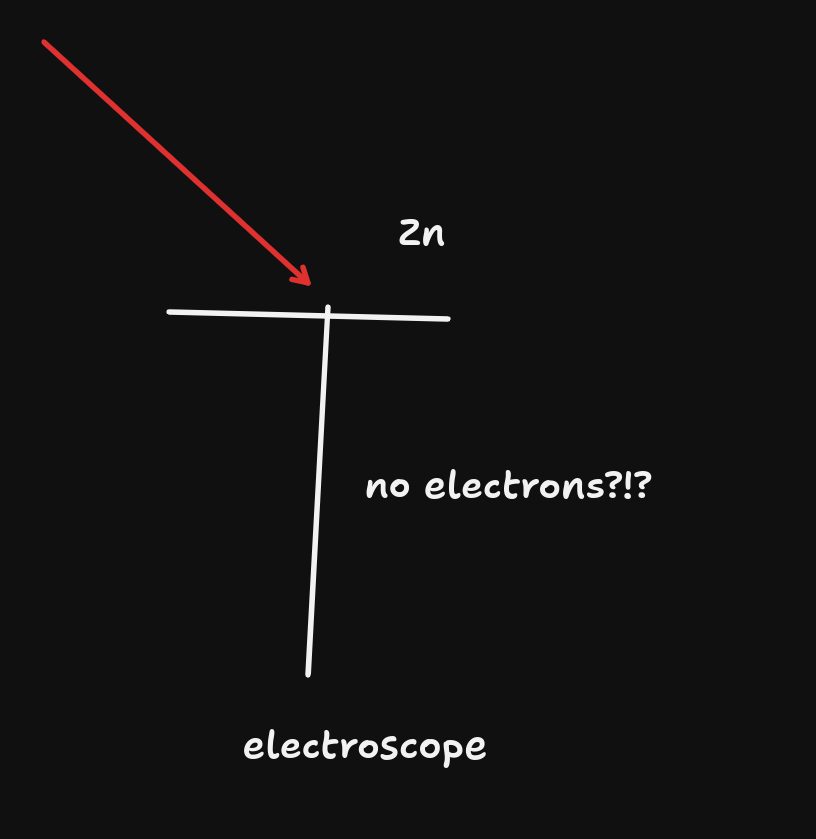
- However, if we use red light (lower frequency), we will not have the electroscope being discharged.
- Even if we increased the intensity up by a lot, we still don't have the electroscope being discharged

Wave properties
- Intensity of a wave is associated with its amplitude.
- However, the photoelectric effect suggests that its not intensity, but frequency that determines whether electrons are dislodged.
Hence, the photoelectric effect is where light acts as a particle.
- UV particles carry more energy per particle (UV has a higher frequency than red light)($E=hf$)
- The higher the frequency, the more quantised packets of energy are delivered.
Photoelectric but better (experiment)
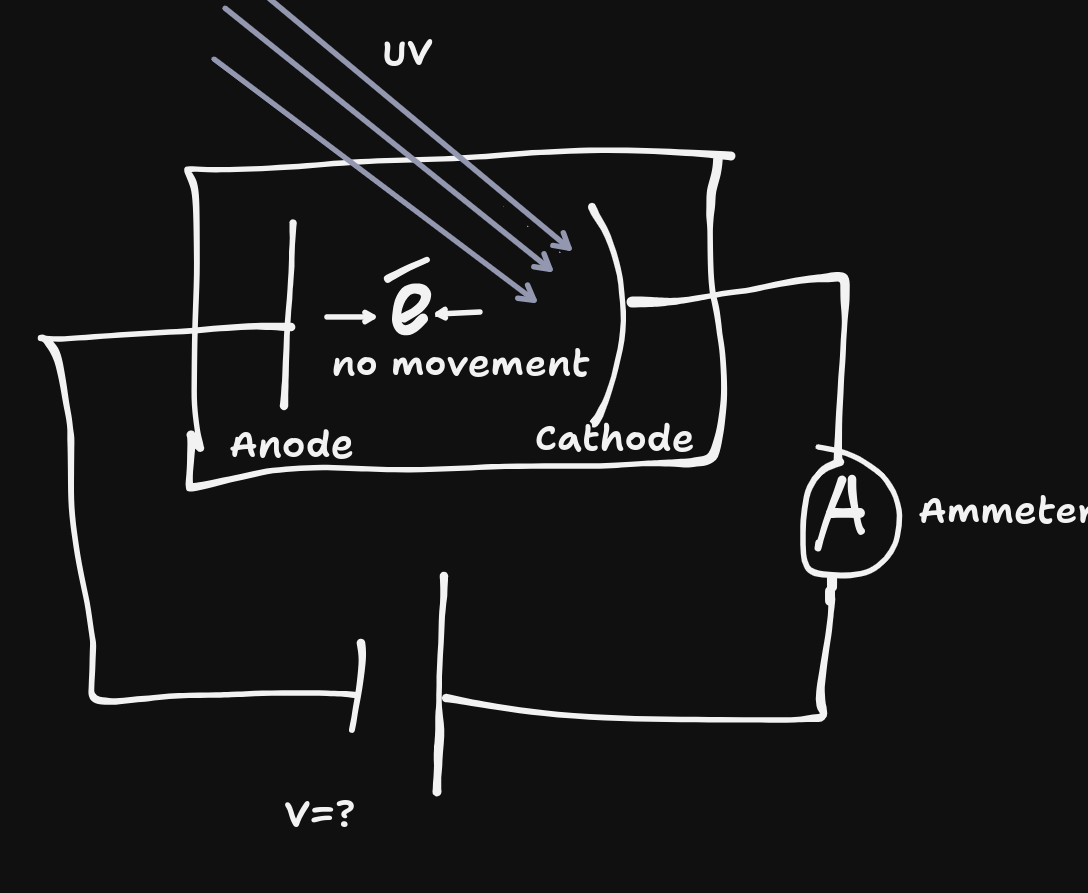
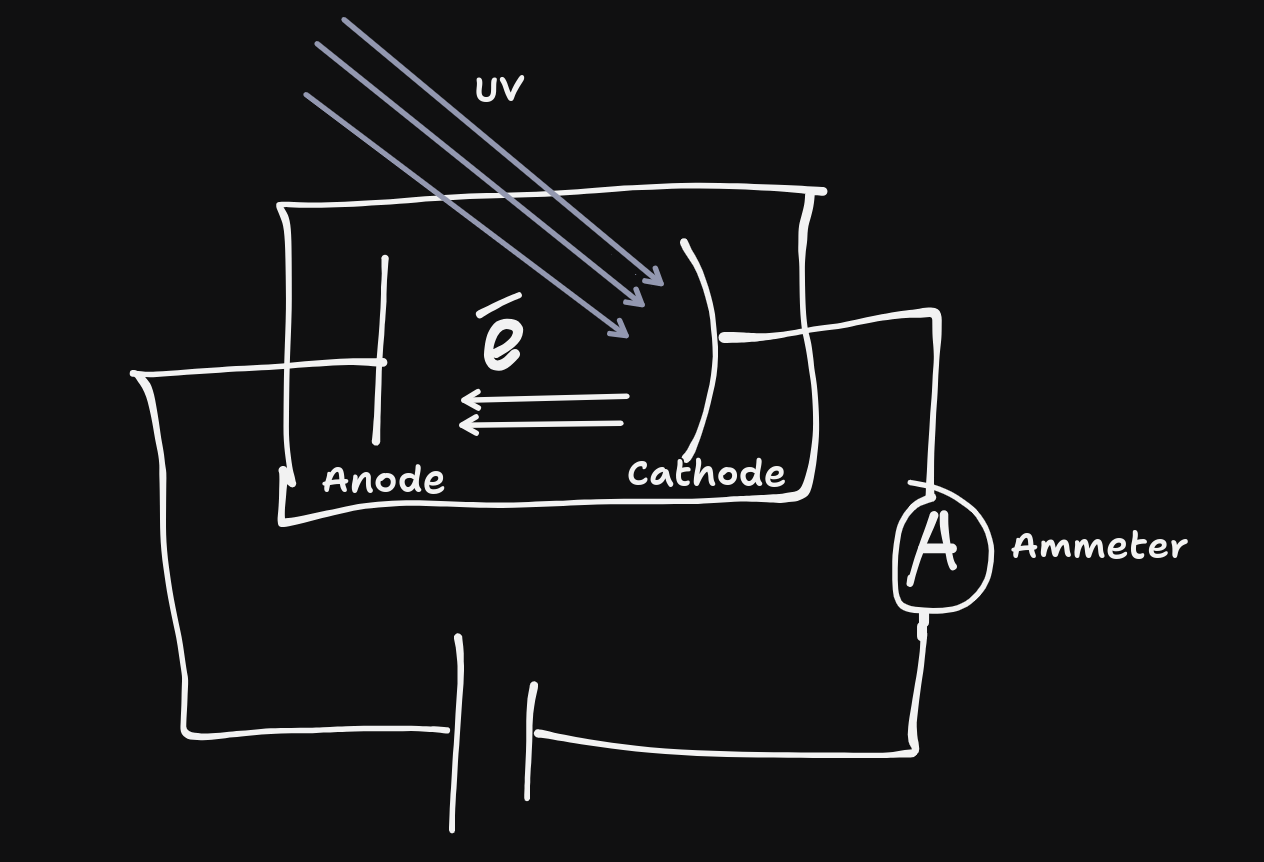 Different frequencies - system conducts electricity (current is created) when light is shone above a certain frequency (Threshold frequency($f_{0}$)), which is different for different metals.
Different frequencies - system conducts electricity (current is created) when light is shone above a certain frequency (Threshold frequency($f_{0}$)), which is different for different metals.
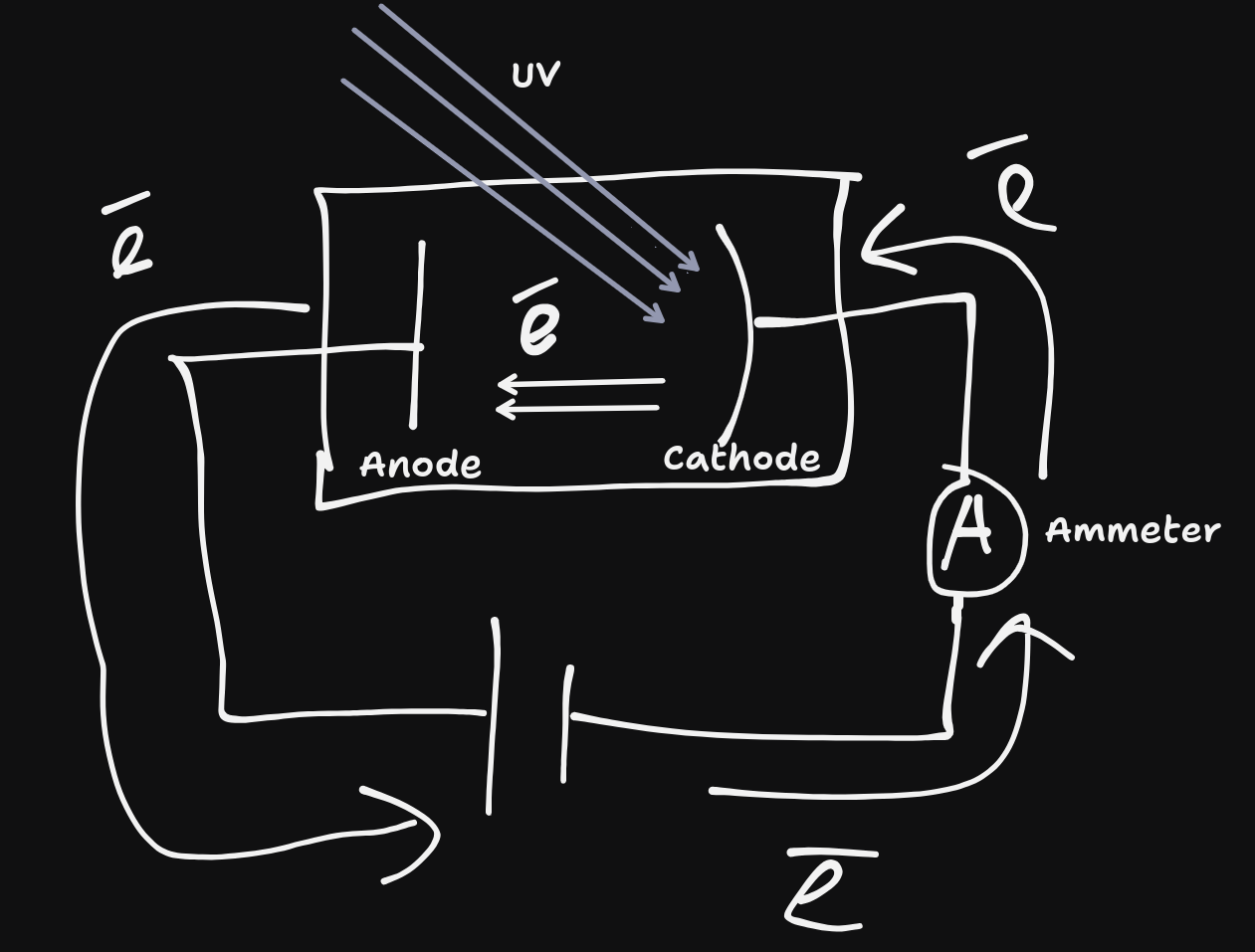
By reversing the terminals and turning up the voltage, eventually the current produced from the photoelectric effect will be stopped. This is called the stopping voltage ($V_{o}$.
As a result:
- E.M radiation can be considered as small packets of energy, i.e. quanta, i.e. photons.
- Hence, $E=hf$, where $h$ is the Planck constant ($6.63\times 10^{-34}Js$)
Past test question
- $2W$ of red light at $600nm$.
- How many photons must be emitted per second to get this power?
$$\begin{align}
E=hf,c=f\lambda \\
\therefore E=\frac{hc}{\lambda} \\
=\frac{6.63\times 10^{-34}\times 3 \times 10^8}{600\times 10^{-9}} \\
=3.32\times 10^{-19}J \\
P=\frac{E}{t}\therefore E=Pt = 2 \times 1 = 2J \\
\therefore \text{n(Photons)}=\frac{2}{3.32\times 10^{-19}} \\
=6.03\times 10^{18}
\end{align}
$$
Extension
$$\begin{align}
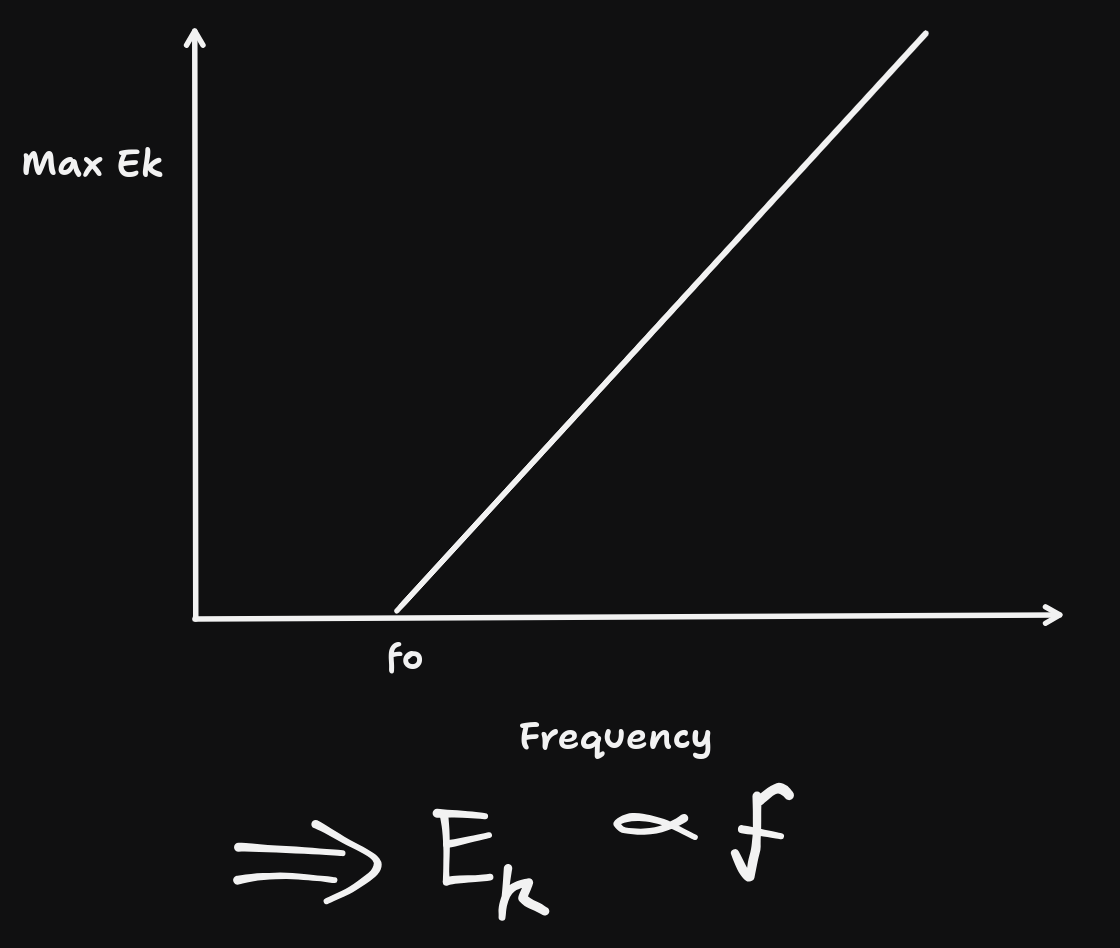
\text{As }E&=hf, \\
\text{Max } E_{k}&=hf-W \\
\text{When }f=&f_{0}, \text{ }Ek=0 \\
\therefore 0&=hf_{0}-W \\
W&=hf_{0} \\
\text{But }W&=Vq \\
\therefore f_{0}&=\frac{Vq}{h}
\end{align}
$$



 Different frequencies - system conducts electricity (current is created) when light is shone above a certain frequency (Threshold frequency($f_{0}$)), which is different for different metals.
Different frequencies - system conducts electricity (current is created) when light is shone above a certain frequency (Threshold frequency($f_{0}$)), which is different for different metals.

