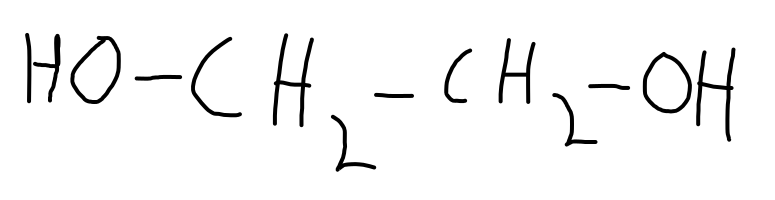Left-click: rotate, Mouse-wheel/middle-click: zoom, Right-click: pan, Escape: close
Condensation
Polyester
- Generally, for a condensation reaction to occur we need a di-acid and a di-alcohol.
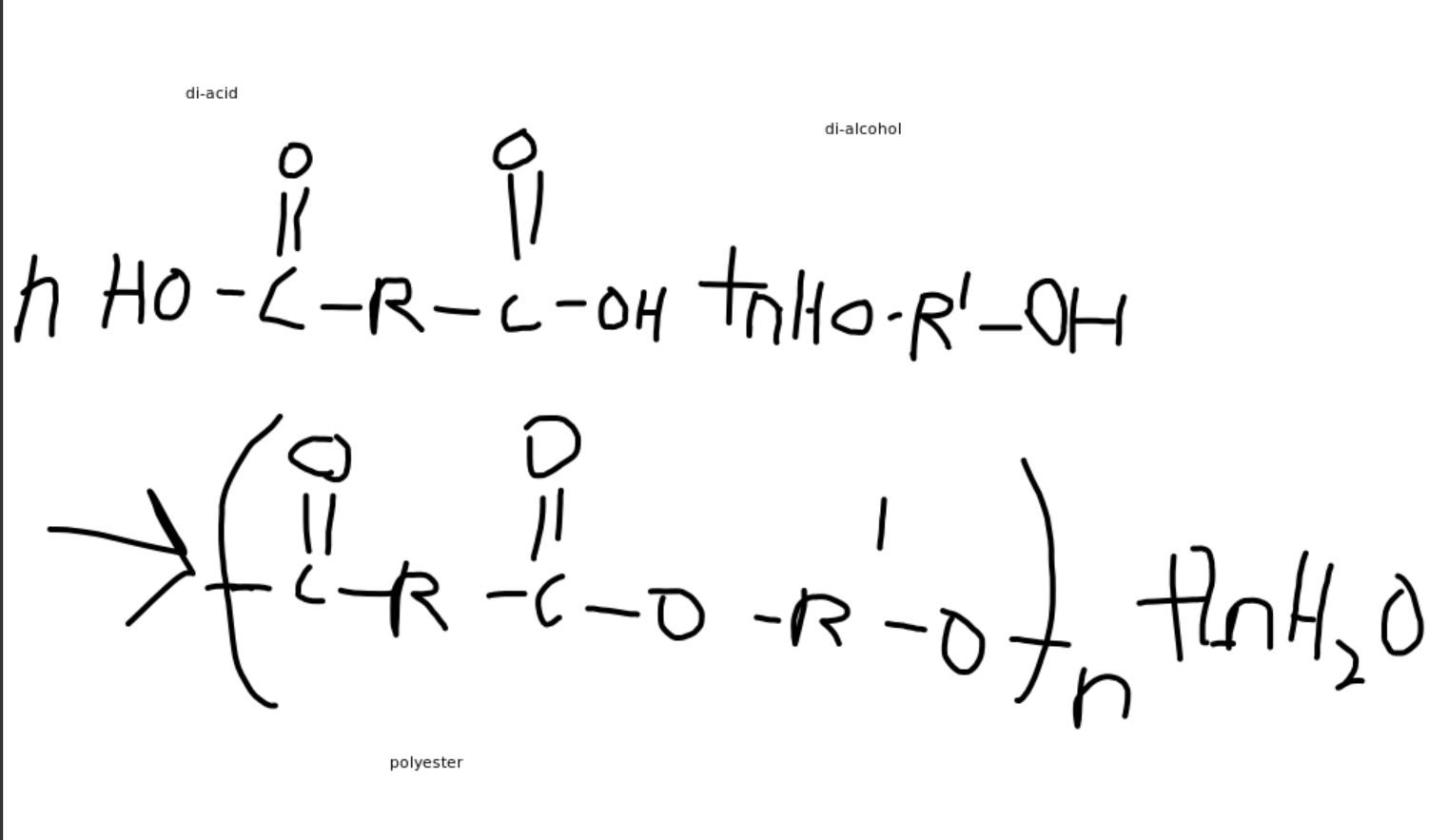
Note that the -OH from the acid and the H from the -OH alcohol group make up the resultant water.
- Large dispersion forces, stronger, lasts longer (fr?)
Polyamide
- Same as polyester formation, but instead is di-acid + di-amine!
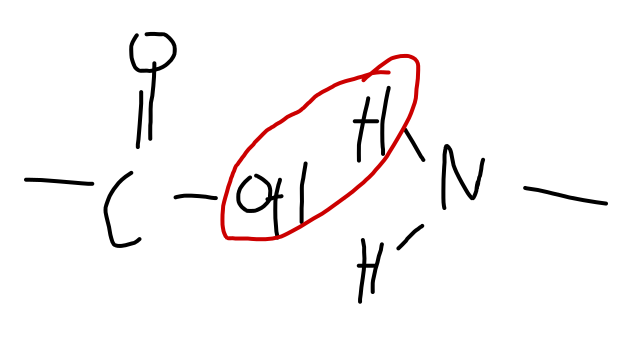
Examples
- Polyethylene terephthalate
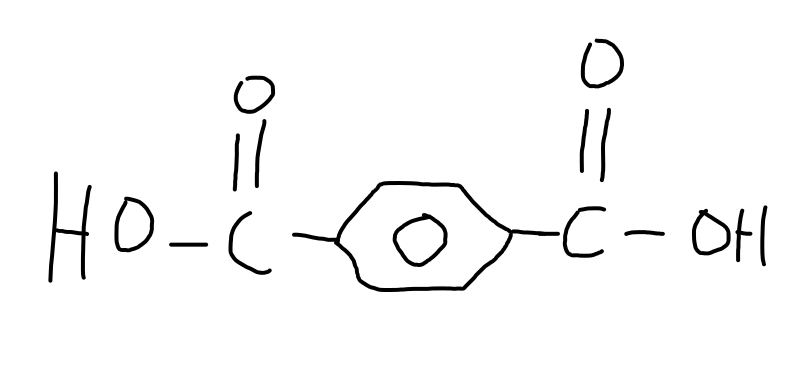 terephthalic acid
terephthalic acid
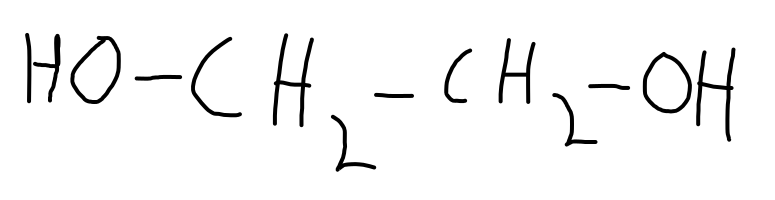
ethan-1,2-diol
-
Benzene ring - lots of electrons
- Increases dispersion forces for strength! (increased M.P, increased B.P)
-
Fibres for clothing, containers for liquids/foods, thermosetting for manufacturing, + glass $\implies$ resins
-
Nylon (6,6) $\implies$ 1st is carbons in amine, 2nd is carbons in acid
-
hexanedioic acid (adipic acid), hex-1,6-diamine
-
Resultant polymer has strong polarity (hydrogen bonds) near peptide linkage.
- N $\delta-$, H $\delta+$, O $\delta-$
- This allows nylon to hydrogen bond, even with itself!!!!! You can line up the molecule, producing strong hydrogen bonds between them
- $\therefore$ high M.P, high B.P

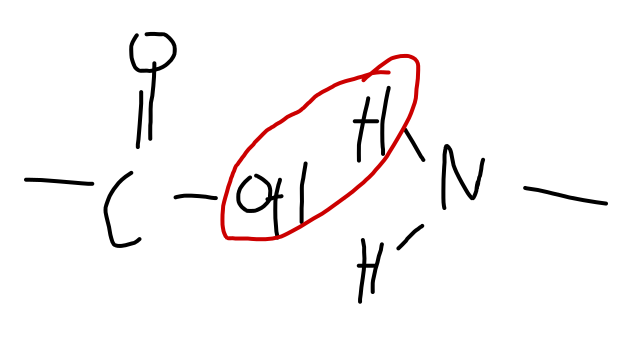
 terephthalic acid
terephthalic acid