Left-click: rotate, Mouse-wheel/middle-click: zoom, Right-click: pan, Escape: close
The following are products of the oxidation of alcohols.
Carboxylic acids
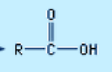 (functional group)
e.g. $CH_{3}COOH$
Hydrogen bonding between molecules:
(functional group)
e.g. $CH_{3}COOH$
Hydrogen bonding between molecules:- Relatively high M.P/B.P. Soluble in water.
- There exists two oxygens on the carboxylic acid that can attract hydrogens (this results in a hydrogen bond, although the double bonded oxygen should be a little weaker
Aldehydes:
- Slightly polar, medium M.P./B.P.? Not particularly soluble
- Cannot hydrogen bond with itself (no H). BUT CAN form hydrogen bonds with water! i.e. H from water can bond to the double bonded O. This O is known as a receptor
Ketones:
- Similar properties to aldehydes,
probably less since disp forces weaker since distribution of electrons in less efficient. This was an incorrect assumption. Apparently, due to some wacky electron distribution rubbish, ketones will have a lower boiling point for whatever reason. This is so stupid. See this wacky discussion
Alcohols:
- Primary alcohols: when the hydroxyl (that's what the $-OH$ is) is on the end, i.e. when the hydroxyl group is bonded to a carbon that is bonded to one(or no) other carbon.
- Secondary alcohols: when the hydroxyl group is bonded to a carbon that is bonded to two other carbons.
- Tertiary alcohols: when the hydroxyl group is bonded to a carbon that is bonded to three other carbons.
Properties
- Hydrogen bonding between molecules - relatively high M.P/B.P. Soluble in water. (separate by distillation(wacky))
Oxidation processes
These process all contain oxidation half-reactions. For these processes to occur, we need something to reduce.
Example organic redox
- Ethanol shaken with excess acidified permanganate.
$RED: 4\times MnO_{4}^{-} + 8H^{+} + 5\overline{e} \to Mn^{2+} + 4H_{2}O$
$OX: 5\times H_{2}O + C_{2}H_{5}OH \to CH_{3}COOH + 4H^{+} + 4\overline{e}$
$\therefore 4MnO_{4}^{-} + 5H_{2}O + 5C_{2}H_{5}OH + 32H^{+} \to 5CH_{3}COOH + 20H^{+} + 4Mn^{2+} + 16H_{2}O$
$\therefore 4MnO_{4}^{-} + 5C_{2}H_{5}OH + 12H^{+} \to 5CH_{3}COOH + 4Mn^{2+} + 11H_{2}O \checkmark$
 (functional group)
e.g. $CH_{3}COOH$
Hydrogen bonding between molecules:
(functional group)
e.g. $CH_{3}COOH$
Hydrogen bonding between molecules: