This is data from an experiment, with some images ported from Notion. I was sadly a Notion user for a long time :(
Practical 1 - Homogenous and Heterogenous mixtures:
Page 10 STAWA Book
Procedure:
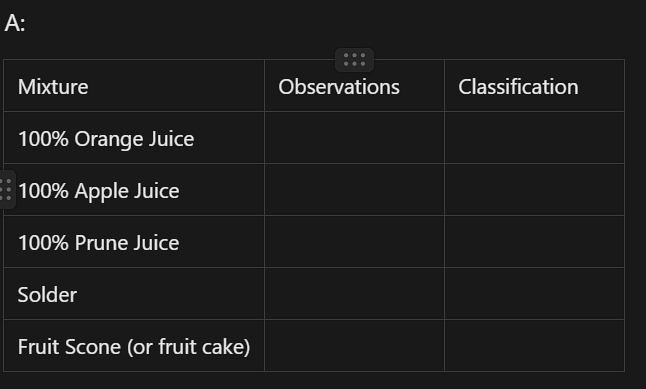 C:
C:
Test the solubility of a small sample (half a teaspoon) of ethanol, charcoal, sodium chloride and copper (II) chloride in separate 15 mL lots of distilled water. Record your observations and classify each sample as soluble or insoluble in the ‘water(solvent)’ column of a table similar to that shown.
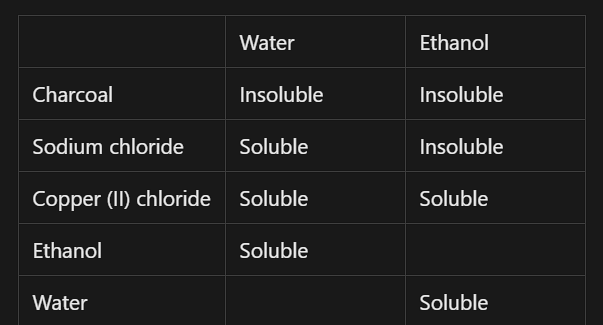
Use solubility to separate different solids. If you find a solvent which dissolves one but not the other, you can add it to the solids, the one that dissolves will create a solution - note this is called filtrate - and the one that does not will stay solid. Then you can use filtration methods and then crystalize the solid in the solution.
Note - research physical properties